JP2005199267A - Metal carrier and method for manufacturing the same - Google Patents
Metal carrier and method for manufacturing the same Download PDFInfo
- Publication number
- JP2005199267A JP2005199267A JP2004362711A JP2004362711A JP2005199267A JP 2005199267 A JP2005199267 A JP 2005199267A JP 2004362711 A JP2004362711 A JP 2004362711A JP 2004362711 A JP2004362711 A JP 2004362711A JP 2005199267 A JP2005199267 A JP 2005199267A
- Authority
- JP
- Japan
- Prior art keywords
- metal
- nanocolloid
- particles
- substrate
- metal nanocolloid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 229910052751 metal Inorganic materials 0.000 title claims abstract description 161
- 239000002184 metal Substances 0.000 title claims abstract description 161
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 40
- 238000000034 method Methods 0.000 title claims abstract description 40
- 239000002245 particle Substances 0.000 claims abstract description 109
- 239000000758 substrate Substances 0.000 claims abstract description 57
- 239000000084 colloidal system Substances 0.000 claims abstract description 34
- 230000001681 protective effect Effects 0.000 claims abstract description 31
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 30
- 238000004070 electrodeposition Methods 0.000 claims abstract description 18
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 67
- 239000007788 liquid Substances 0.000 claims description 39
- 229910052697 platinum Inorganic materials 0.000 claims description 33
- 239000000463 material Substances 0.000 claims description 24
- 229910000510 noble metal Inorganic materials 0.000 claims description 24
- 239000003638 chemical reducing agent Substances 0.000 claims description 15
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 14
- 229910044991 metal oxide Inorganic materials 0.000 claims description 10
- 150000004706 metal oxides Chemical class 0.000 claims description 10
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 claims description 8
- 229910052707 ruthenium Inorganic materials 0.000 claims description 8
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 7
- 239000003575 carbonaceous material Substances 0.000 claims description 7
- 229910052763 palladium Inorganic materials 0.000 claims description 7
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 claims description 6
- 229910052737 gold Inorganic materials 0.000 claims description 6
- 239000010931 gold Substances 0.000 claims description 6
- 239000007769 metal material Substances 0.000 claims description 6
- 229910052703 rhodium Inorganic materials 0.000 claims description 6
- 239000010948 rhodium Substances 0.000 claims description 6
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 claims description 6
- 229910052762 osmium Inorganic materials 0.000 claims description 5
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 claims description 5
- 229910052702 rhenium Inorganic materials 0.000 claims description 5
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 claims description 5
- 229920000620 organic polymer Polymers 0.000 claims description 4
- 229910052799 carbon Inorganic materials 0.000 claims description 3
- 229910010293 ceramic material Inorganic materials 0.000 claims description 3
- 239000002861 polymer material Substances 0.000 claims description 3
- 239000006185 dispersion Substances 0.000 abstract description 15
- 230000008569 process Effects 0.000 abstract description 3
- 239000000243 solution Substances 0.000 description 36
- 239000003054 catalyst Substances 0.000 description 23
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 20
- 239000000446 fuel Substances 0.000 description 14
- 239000001509 sodium citrate Substances 0.000 description 13
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 13
- 239000010410 layer Substances 0.000 description 12
- 229920001955 polyphenylene ether Polymers 0.000 description 12
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 10
- 239000001301 oxygen Substances 0.000 description 10
- 229910052760 oxygen Inorganic materials 0.000 description 10
- -1 superoxide anions Chemical class 0.000 description 10
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- 238000000354 decomposition reaction Methods 0.000 description 8
- 235000019441 ethanol Nutrition 0.000 description 8
- 239000010409 thin film Substances 0.000 description 8
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 7
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 7
- 229910045601 alloy Inorganic materials 0.000 description 7
- 239000000956 alloy Substances 0.000 description 7
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid group Chemical class C(CC(O)(C(=O)O)CC(=O)O)(=O)O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 7
- 239000010419 fine particle Substances 0.000 description 7
- 150000002430 hydrocarbons Chemical class 0.000 description 7
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 7
- 238000002407 reforming Methods 0.000 description 7
- 150000003839 salts Chemical class 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 6
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- 230000002776 aggregation Effects 0.000 description 6
- 238000004220 aggregation Methods 0.000 description 6
- 150000001298 alcohols Chemical class 0.000 description 6
- 238000009835 boiling Methods 0.000 description 6
- 238000000576 coating method Methods 0.000 description 6
- 239000007789 gas Substances 0.000 description 6
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 6
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 230000003197 catalytic effect Effects 0.000 description 5
- 235000015165 citric acid Nutrition 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- 229930195733 hydrocarbon Natural products 0.000 description 5
- 239000003456 ion exchange resin Substances 0.000 description 5
- 229920003303 ion-exchange polymer Polymers 0.000 description 5
- 230000001699 photocatalysis Effects 0.000 description 5
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 239000000919 ceramic Substances 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 150000002170 ethers Chemical class 0.000 description 4
- 239000010408 film Substances 0.000 description 4
- 239000001257 hydrogen Substances 0.000 description 4
- 229910052739 hydrogen Inorganic materials 0.000 description 4
- 150000002500 ions Chemical class 0.000 description 4
- 150000002576 ketones Chemical class 0.000 description 4
- TZIHFWKZFHZASV-UHFFFAOYSA-N methyl formate Chemical compound COC=O TZIHFWKZFHZASV-UHFFFAOYSA-N 0.000 description 4
- 238000006722 reduction reaction Methods 0.000 description 4
- 239000011347 resin Substances 0.000 description 4
- 229920005989 resin Polymers 0.000 description 4
- 238000004659 sterilization and disinfection Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- 239000004734 Polyphenylene sulfide Substances 0.000 description 3
- 239000004372 Polyvinyl alcohol Substances 0.000 description 3
- 239000011149 active material Substances 0.000 description 3
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 3
- 150000001735 carboxylic acids Chemical class 0.000 description 3
- 239000007809 chemical reaction catalyst Substances 0.000 description 3
- 239000004020 conductor Substances 0.000 description 3
- 239000000645 desinfectant Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000007772 electroless plating Methods 0.000 description 3
- 239000000835 fiber Substances 0.000 description 3
- 229910052809 inorganic oxide Inorganic materials 0.000 description 3
- 239000011941 photocatalyst Substances 0.000 description 3
- 229920000069 polyphenylene sulfide Polymers 0.000 description 3
- 229920002451 polyvinyl alcohol Polymers 0.000 description 3
- 239000012429 reaction media Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 239000004094 surface-active agent Substances 0.000 description 3
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 2
- 229910000831 Steel Inorganic materials 0.000 description 2
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 150000001342 alkaline earth metals Chemical class 0.000 description 2
- 238000004061 bleaching Methods 0.000 description 2
- 239000007844 bleaching agent Substances 0.000 description 2
- 239000006229 carbon black Substances 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 229920001429 chelating resin Polymers 0.000 description 2
- 239000010941 cobalt Substances 0.000 description 2
- 229910017052 cobalt Inorganic materials 0.000 description 2
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 239000002612 dispersion medium Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 229910002804 graphite Inorganic materials 0.000 description 2
- 239000010439 graphite Substances 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 229910021645 metal ion Inorganic materials 0.000 description 2
- 239000002923 metal particle Substances 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 239000003345 natural gas Substances 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- 229910001453 nickel ion Inorganic materials 0.000 description 2
- 125000000962 organic group Chemical group 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 239000003208 petroleum Substances 0.000 description 2
- 238000005240 physical vapour deposition Methods 0.000 description 2
- 150000003057 platinum Chemical class 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000005518 polymer electrolyte Substances 0.000 description 2
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010959 steel Substances 0.000 description 2
- 230000001954 sterilising effect Effects 0.000 description 2
- 239000004408 titanium dioxide Substances 0.000 description 2
- 229920003169 water-soluble polymer Polymers 0.000 description 2
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- FFJCNSLCJOQHKM-CLFAGFIQSA-N (z)-1-[(z)-octadec-9-enoxy]octadec-9-ene Chemical compound CCCCCCCC\C=C/CCCCCCCCOCCCCCCCC\C=C/CCCCCCCC FFJCNSLCJOQHKM-CLFAGFIQSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- 229920000178 Acrylic resin Polymers 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Natural products CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 1
- 229910001018 Cast iron Inorganic materials 0.000 description 1
- 229920000298 Cellophane Polymers 0.000 description 1
- 229920002284 Cellulose triacetate Polymers 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 229910000640 Fe alloy Inorganic materials 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- XOBKSJJDNFUZPF-UHFFFAOYSA-N Methoxyethane Chemical compound CCOC XOBKSJJDNFUZPF-UHFFFAOYSA-N 0.000 description 1
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 1
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 1
- VEQPNABPJHWNSG-UHFFFAOYSA-N Nickel(2+) Chemical compound [Ni+2] VEQPNABPJHWNSG-UHFFFAOYSA-N 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 239000004696 Poly ether ether ketone Substances 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004697 Polyetherimide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 229920001328 Polyvinylidene chloride Polymers 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- 229910021536 Zeolite Inorganic materials 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- SMEGJBVQLJJKKX-HOTMZDKISA-N [(2R,3S,4S,5R,6R)-5-acetyloxy-3,4,6-trihydroxyoxan-2-yl]methyl acetate Chemical compound CC(=O)OC[C@@H]1[C@H]([C@@H]([C@H]([C@@H](O1)O)OC(=O)C)O)O SMEGJBVQLJJKKX-HOTMZDKISA-N 0.000 description 1
- NNLVGZFZQQXQNW-ADJNRHBOSA-N [(2r,3r,4s,5r,6s)-4,5-diacetyloxy-3-[(2s,3r,4s,5r,6r)-3,4,5-triacetyloxy-6-(acetyloxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6s)-4,5,6-triacetyloxy-2-(acetyloxymethyl)oxan-3-yl]oxyoxan-2-yl]methyl acetate Chemical compound O([C@@H]1O[C@@H]([C@H]([C@H](OC(C)=O)[C@H]1OC(C)=O)O[C@H]1[C@@H]([C@@H](OC(C)=O)[C@H](OC(C)=O)[C@@H](COC(C)=O)O1)OC(C)=O)COC(=O)C)[C@@H]1[C@@H](COC(C)=O)O[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O NNLVGZFZQQXQNW-ADJNRHBOSA-N 0.000 description 1
- 235000011054 acetic acid Nutrition 0.000 description 1
- 229940081735 acetylcellulose Drugs 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 230000009172 bursting Effects 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 239000003990 capacitor Substances 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 239000003610 charcoal Substances 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 239000011247 coating layer Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 230000000249 desinfective effect Effects 0.000 description 1
- 229920005994 diacetyl cellulose Polymers 0.000 description 1
- HNPSIPDUKPIQMN-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Al]O[Al]=O HNPSIPDUKPIQMN-UHFFFAOYSA-N 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 238000009713 electroplating Methods 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 229910003437 indium oxide Inorganic materials 0.000 description 1
- PJXISJQVUVHSOJ-UHFFFAOYSA-N indium(iii) oxide Chemical compound [O-2].[O-2].[O-2].[In+3].[In+3] PJXISJQVUVHSOJ-UHFFFAOYSA-N 0.000 description 1
- 239000011810 insulating material Substances 0.000 description 1
- 229910052741 iridium Inorganic materials 0.000 description 1
- GKOZUEZYRPOHIO-UHFFFAOYSA-N iridium atom Chemical compound [Ir] GKOZUEZYRPOHIO-UHFFFAOYSA-N 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 238000010030 laminating Methods 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000003949 liquefied natural gas Substances 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229910001510 metal chloride Inorganic materials 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 238000006864 oxidative decomposition reaction Methods 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 238000001637 plasma atomic emission spectroscopy Methods 0.000 description 1
- 239000002574 poison Substances 0.000 description 1
- 231100000614 poison Toxicity 0.000 description 1
- 229920003207 poly(ethylene-2,6-naphthalate) Polymers 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920002492 poly(sulfone) Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920001707 polybutylene terephthalate Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 229920001225 polyester resin Polymers 0.000 description 1
- 239000004645 polyester resin Substances 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920002530 polyetherether ketone Polymers 0.000 description 1
- 229920001601 polyetherimide Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000011112 polyethylene naphthalate Substances 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229920000306 polymethylpentene Polymers 0.000 description 1
- 239000011116 polymethylpentene Substances 0.000 description 1
- 229920005672 polyolefin resin Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 239000005033 polyvinylidene chloride Substances 0.000 description 1
- 239000001508 potassium citrate Substances 0.000 description 1
- 229960002635 potassium citrate Drugs 0.000 description 1
- QEEAPRPFLLJWCF-UHFFFAOYSA-K potassium citrate (anhydrous) Chemical compound [K+].[K+].[K+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O QEEAPRPFLLJWCF-UHFFFAOYSA-K 0.000 description 1
- 235000011082 potassium citrates Nutrition 0.000 description 1
- 239000010970 precious metal Substances 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 238000000629 steam reforming Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 239000011135 tin Substances 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- YWYZEGXAUVWDED-UHFFFAOYSA-N triammonium citrate Chemical compound [NH4+].[NH4+].[NH4+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O YWYZEGXAUVWDED-UHFFFAOYSA-N 0.000 description 1
- HRXKRNGNAMMEHJ-UHFFFAOYSA-K trisodium citrate Chemical group [Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O HRXKRNGNAMMEHJ-UHFFFAOYSA-K 0.000 description 1
- 238000007740 vapor deposition Methods 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
- 239000010457 zeolite Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Abstract
Description
本発明は、金属担持体の製造方法及び金属担持体に関する。さらに詳しくは、本発明は保護コロイド形成剤を実質上含まず、しかも平均粒径1〜20nm程度の金属ナノコロイド粒子を、比較的高濃度で含有しても分散安定性が良好な金属ナノコロイド液を用い、電着法により金属ナノコロイド粒子を基体に、高速かつ高濃度に効率よく担持させ、各種分野で有用な金属担持体を製造する方法、及びこの方法で得られた金属担持体に関するものである。 The present invention relates to a method for producing a metal carrier and a metal carrier. More specifically, the present invention is substantially free of a protective colloid-forming agent, and has good dispersion stability even if it contains metal nanocolloid particles having an average particle size of about 1 to 20 nm at a relatively high concentration. The present invention relates to a method for producing a metal carrier useful in various fields by efficiently supporting metal nanocolloid particles on a substrate at high speed and high concentration by electrodeposition using a liquid, and a metal carrier obtained by this method Is.
近年、炭素系材料、セラミックス・金属酸化物系材料、金属系材料、又は有機高分子系材料などの基体に、金属を担持させてなる金属担持体は、機能性材料として、各種分野において注目され、新しい用途の開発研究が積極的になされている。
前記金属担持体としては、例えば(1)樹脂粒子などの絶縁性粒子表面に貴金属を被覆してなる導電性粒子、(2)樹脂材料の表面に導電性を付与し、さらに貴金属を担持してなる消毒薬や漂白剤の分解触媒、(3)多孔質担体に貴金属を担持させてなる自動車排気ガスの浄化用触媒、(4)高触媒機能を有する薄膜上に貴金属被覆を設けてなる光触媒薄膜などが開発されている。さらに、燃料電池分野においては、(5)無機酸化物担体にルテニウムなどの貴金属を担持させてなる、炭化水素化合物や酸素含有炭化水素化合物を改質して水素を発生させるための改質触媒、(6)水素ガス中の一酸化炭素を低減させる、無機酸化物担体に貴金属が担持されたシフト反応触媒、(7)炭素系材料に貴金属を担持してなる燃料電池用電極触媒などが開発されている。
In recent years, metal supports in which a metal is supported on a substrate such as a carbon-based material, a ceramic / metal oxide-based material, a metal-based material, or an organic polymer-based material have attracted attention in various fields as a functional material. Research and development of new applications has been actively conducted.
Examples of the metal carrier include (1) conductive particles obtained by coating the surface of insulating particles such as resin particles with noble metal, and (2) imparting conductivity to the surface of the resin material, and further carrying the noble metal. A disinfectant and bleach decomposition catalyst, (3) a catalyst for purifying automobile exhaust gas by supporting a noble metal on a porous carrier, and (4) a photocatalytic thin film having a noble metal coating on a thin film having a high catalytic function Etc. are being developed. Further, in the field of fuel cells, (5) a reforming catalyst for generating hydrogen by reforming a hydrocarbon compound or an oxygen-containing hydrocarbon compound formed by supporting a noble metal such as ruthenium on an inorganic oxide carrier, (6) Shift reaction catalysts in which noble metals are supported on inorganic oxide supports that reduce carbon monoxide in hydrogen gas, (7) Electrode catalysts for fuel cells in which noble metals are supported on carbon-based materials, etc. have been developed. ing.
次に、前記各金属担持体の背景について説明する。
〔導電性粒子〕
液晶などの電極部には、圧着によって導電性粒子を変形させ、特定の電極間又は方向で通電させる異方性導電部材が用いられており、この異方性導電部材用に、樹脂粒子などの絶縁粒子表面に金などを被覆した導電性粒子が用いられる。また、電子部品に用いられるインダクター、積層コンデンサーは、導電体層に磁性体層を積層し、一体焼結することにより作製されるが、前記導電体層の形成には、通常導電性粒子を含む導電体形成用ペーストが用いられる。
Next, the background of each metal carrier will be described.
[Conductive particles]
An electrode member such as a liquid crystal uses an anisotropic conductive member that deforms conductive particles by pressure bonding and energizes between specific electrodes or in a direction. For this anisotropic conductive member, resin particles or the like are used. Conductive particles whose surfaces are coated with gold or the like are used. Inductors and multilayer capacitors used in electronic parts are manufactured by laminating a magnetic layer on a conductor layer and sintering it integrally. However, the conductor layer usually contains conductive particles. A conductor forming paste is used.
〔消毒薬の分解触媒〕
過酸化水素、オゾンなどの過酸化物は、消毒、殺菌及び漂白作用を有し、有用に用いられている物質である。しかし、高濃度では人体にも有害であり、悪影響を及ぼすことがあるため、高濃度の状態で使用して、目的の効果を得た後には中和分解処理を施すことが行われる。この分解処理において、白金などの貴金属が分解触媒として作用することが知られている。例えば、ポリフェニレンエーテル(以下「PPE」と略することがある)やポリフェニレンサルファイド(以下「PPS」と略することがある)等の樹脂材料の表面にあらかじめ導電性を付与し、これに貴金属材料を担持することによって、軽量かつ多様な構造を有する消毒薬、漂白剤等の分解触媒材料を製造することができる。
[Disinfection decomposition catalyst]
Peroxides such as hydrogen peroxide and ozone have a disinfecting, sterilizing and bleaching action and are useful materials. However, since it is harmful to the human body at a high concentration and may have an adverse effect, neutralization and decomposition treatment is performed after obtaining the desired effect when used in a high concentration state. In this decomposition treatment, it is known that noble metals such as platinum act as decomposition catalysts. For example, the surface of a resin material such as polyphenylene ether (hereinafter sometimes abbreviated as “PPE”) or polyphenylene sulfide (hereinafter sometimes abbreviated as “PPS”) is given conductivity in advance, and a noble metal material is added thereto. By carrying it, it is possible to produce decomposition catalyst materials such as disinfectants and bleaching agents having light weight and various structures.
〔自動車排気ガスの浄化用触媒〕
近年、リーンバーン自動車排ガスの浄化用触媒として、NOx吸蔵還元型触媒が広く使用されている。このNOx吸蔵還元型触媒は白金、パラジウム等の触媒活性を有する貴金属粒子と、主にバリウム等のアルカリ土類金属の炭酸塩を、アルミナ、ジルコニア等のセラミックのペレット状、又はハニカム成形体若しくはセラミックをコーティングした金属ハニカムといった多孔質体である担体上に担持されたものである。このNOx吸蔵還元型触媒において、貴金属粒子はNOxの分解を促進するための触媒成分として作用し、一方のアルカリ土類金属はNOxの吸蔵剤としての役割を有するものである。
[Automobile exhaust gas purification catalyst]
In recent years, NOx occlusion reduction type catalysts have been widely used as catalysts for purifying lean burn automobile exhaust gas. This NOx occlusion reduction type catalyst is composed of noble metal particles having catalytic activity such as platinum and palladium and carbonates of alkaline earth metals such as barium, ceramic pellets such as alumina and zirconia, or honeycomb molded bodies or ceramics. Is supported on a carrier which is a porous body such as a metal honeycomb coated with bismuth. In this NOx occlusion reduction type catalyst, the noble metal particles act as a catalyst component for promoting the decomposition of NOx, and one alkaline earth metal has a role as a NOx occlusion agent.
〔光触媒薄膜〕
光触媒活性材料(以下、単に光触媒と称することがある。)は、そのバンドギャップ以上のエネルギーの光を照射すると、励起されて伝導帯に電子が生じ、かつ価電子帯に正孔が生じる。そして、生成した電子は表面酸素を還元してスーパーオキサイドアニオン(・O2-)を生成させると共に、正孔は表面水酸基を酸化して水酸ラジカル(・OH)を生成し、これらの反応性活性酸素種が強い酸化分解機能を発揮し、光触媒の表面に付着している有機物質を高効率で分解することが知られている。
この光触媒活性材料としては、二酸化チタン、特にアナターゼ型二酸化チタンが実用的なものとして有用である。そして、この光触媒活性材料層には、光触媒活性を促進させる目的で白金、パラジウム、ロジウム、ルテニウムなどの白金族金属の被覆層を設けることが行われている。
[Photocatalytic thin film]
A photocatalytically active material (hereinafter sometimes simply referred to as a photocatalyst) is excited to generate electrons in the conduction band and holes in the valence band when irradiated with light having energy higher than the band gap. The generated electrons reduce surface oxygen to generate superoxide anions (• O 2− ), and holes oxidize surface hydroxyl groups to generate hydroxyl radicals (• OH). It is known that active oxygen species exert a strong oxidative decomposition function and decompose organic substances adhering to the surface of the photocatalyst with high efficiency.
As this photocatalytically active material, titanium dioxide, particularly anatase type titanium dioxide, is useful as a practical material. The photocatalytically active material layer is provided with a coating layer of a platinum group metal such as platinum, palladium, rhodium or ruthenium for the purpose of promoting photocatalytic activity.
〔燃料電池分野における各金属担持体〕
燃料電池は、水素と酸素を電気化学的に反応させることにより、化学エネルギーを電気エネルギーに変換するものであって、エネルギーの利用効率が高いという特徴を有しており、民生用、産業用あるいは自動車用などとして、実用化研究が積極的になされている。
水素源としては、メタノール、メタンを主体とする液化天然ガス、この天然ガスを主成分とする都市ガス、天然ガスを原料とする合成液体燃料、さらには石油系のLPG、ナフサ、灯油などの石油系炭化水素等の炭化水素系燃料や酸素含有炭化水素系燃料の使用が研究されている。
[Each metal carrier in the fuel cell field]
A fuel cell is a device that converts chemical energy into electric energy by electrochemically reacting hydrogen and oxygen, and has a feature of high energy use efficiency. Research into practical use has been actively conducted for automobiles.
As a hydrogen source, liquefied natural gas mainly composed of methanol and methane, city gas mainly composed of this natural gas, synthetic liquid fuel made from natural gas, and petroleum-based LPG, naphtha, kerosene and other petroleum The use of hydrocarbon fuels such as hydrocarbons and oxygen-containing hydrocarbon fuels has been studied.
前記の炭化水素系燃料や酸素含有炭化水素系燃料を用いて水素を生成させる場合、水蒸気改質や部分酸化改質などの改質処理が施されるが、その際、改質触媒として無機酸化物担体に、ルテニウムなどの貴金属を担持した触媒が一般的に用いられる。
また、前記の改質処理で得られる水素ガス中には、通常COが含まれており、このCOは、燃料電池、特に固体高分子型燃料電池などの低温作動型燃料電池において、電極として用いられる白金触媒を被毒しやすい。したがって、このCOを無害のCO2などに転化して、CO濃度を低減させることが重要となり、そのためシフト反応を利用する方法が通常用いられ、そしてこのシフト反応触媒として、無機酸化物担体に貴金属を担持した触媒が使用される。
When hydrogen is produced using the above-mentioned hydrocarbon fuel or oxygen-containing hydrocarbon fuel, reforming treatment such as steam reforming or partial oxidation reforming is performed. At that time, inorganic oxidation is used as a reforming catalyst. A catalyst in which a noble metal such as ruthenium is supported on a material carrier is generally used.
Further, the hydrogen gas obtained by the above reforming treatment usually contains CO, and this CO is used as an electrode in a fuel cell, particularly in a low temperature operation type fuel cell such as a polymer electrolyte fuel cell. It is easy to poison the platinum catalyst. Therefore, it is important to reduce the CO concentration by converting this CO into harmless CO 2 or the like. Therefore, a method utilizing a shift reaction is usually used, and a noble metal is used as an inorganic oxide support as the shift reaction catalyst. Is used.
さらに、固体高分子型燃料電池の構成要素である電極においては、化学反応を促進させるために、グラファイト、カーボンブラックなどの炭素系材料に白金などの貴金属を担持させたものが用いられている。
このような貴金属担持体は、その用途や基体の種類などに応じて、真空蒸着やスパッタリングなどの物理的気相蒸着法(PVD法)、あるいは電気めっき法、無電解めっき法、金属コロイド担持法などの湿式法の中から適宜選ばれた方法を用いて作製されている。これらの中で、金属コロイド担持法は、金属ナノコロイド微粒子を含む金属ナノコロイド液を用いて、浸漬、噴霧、蒸発乾固などの塗布方法によって、基体に金属ナノコロイド粒子を担持させる方法であり、操作が簡単であって、高価な塗布装置を必要としないなどの利点を有している。なお、ここでナノコロイドとは、径が100nm未満程度のコロイド粒子をいう。
Furthermore, in an electrode that is a constituent element of a polymer electrolyte fuel cell, a material in which a noble metal such as platinum is supported on a carbon-based material such as graphite or carbon black is used in order to promote a chemical reaction.
Such a noble metal carrier may be a physical vapor deposition method (PVD method) such as vacuum vapor deposition or sputtering, or an electroplating method, an electroless plating method, a metal colloid carrying method, depending on the application and type of substrate. It is produced using a method appropriately selected from wet methods such as. Among these, the metal colloid support method is a method of supporting metal nanocolloid particles on a substrate by a coating method such as immersion, spraying, evaporation and drying using a metal nanocolloid liquid containing metal nanocolloid fine particles. The operation is simple, and there is an advantage that an expensive coating apparatus is not required. Here, the nano colloid means colloidal particles having a diameter of less than about 100 nm.
ところが、この金属ナノコロイド液は、一般に金属ナノコロイド粒子の分散安定性が悪く、凝集を生じやすいために、通常ポリビニルアルコール、ポリビニルピロリドン、ゼラチンなどの水溶性高分子化合物、あるいは界面活性剤などの保護コロイド形成剤を加えて、保護コロイドを形成させることにより、金属ナノコロイド粒子の分散安定性を向上させる処置がとられている。
例えば、絶縁性基板上に堆積させた金属酸化物薄膜を、貴金属塩化物水溶液中に、保護コロイド形成剤としてポリエチレングリコールモノオレイルエーテル水溶液を添加することによって得られた貴金属コロイド中に浸漬し、金属酸化物薄膜上に貴金属を担持させる方法(例えば、特許文献1参照)、炭素数1〜4のアルキル基を少なくとも一つ有する4級アンモニウム塩を保護コロイド形成剤として用い、貴金属コロイド液を調製し、これを多孔質担体に吸着させてなる排ガス浄化触媒の製造方法(例えば、特許文献2参照)、光触媒機能を有する薄膜上に、界面活性剤で安定させた貴金属微粒子コロイドを塗布した後、還元性雰囲気中約400〜600℃で加熱処理する貴金属微粒子担持光触媒薄膜の製造法(例えば、特許文献3参照)などが開示されている。
However, since the metal nanocolloid liquid generally has poor dispersion stability of the metal nanocolloid particles and is likely to cause aggregation, water-soluble polymer compounds such as polyvinyl alcohol, polyvinylpyrrolidone and gelatin, or surfactants are usually used. A treatment for improving the dispersion stability of the metal nanocolloid particles has been taken by adding a protective colloid-forming agent to form a protective colloid.
For example, a metal oxide thin film deposited on an insulating substrate is immersed in a noble metal colloid obtained by adding a polyethylene glycol monooleyl ether aqueous solution as a protective colloid forming agent to a noble metal chloride aqueous solution. A method of supporting a noble metal on an oxide thin film (see, for example, Patent Document 1), and using a quaternary ammonium salt having at least one alkyl group having 1 to 4 carbon atoms as a protective colloid forming agent, a noble metal colloid solution is prepared. , A method for producing an exhaust gas purification catalyst by adsorbing it on a porous carrier (see, for example, Patent Document 2), applying a noble metal fine particle colloid stabilized with a surfactant on a thin film having a photocatalytic function, and then reducing For producing a photocatalyst thin film carrying noble metal fine particles by heat treatment at about 400 to 600 ° C. in a neutral atmosphere (see, for example, Patent Document 3) Etc. have been disclosed.
しかしながら、このように保護コロイド形成剤を用いると、基体に金属ナノコロイド粒子を担持させた場合、該コロイド粒子の表面に保護コロイド形成剤が付着したまま担持されるので、得られる金属担持体は有機物質を含むことになる。このような有機物質を含む金属担持体は、目的の機能を十分に発揮し得ないことがあり、その場合には、焼成処理により、有機物質を除く処置が必要となる。さらに、基体の種類によっては、焼成処理が不可能なものがあり、基体の種類が制限されるのを免れないという問題も生じる。
保護コロイド形成剤を用いない金属ナノコロイド液の製造方法としては、例えば金属塩化物が溶解した溶液に還元剤を添加し、金属イオンを還元することで金属微粒子を生成させる方法が知られている(例えば特許文献4、非特許文献1参照)。
However, when the protective colloid-forming agent is used in this way, when the metal nanocolloid particles are supported on the substrate, the protective colloid-forming agent is supported on the surface of the colloidal particles. Contains organic substances. A metal carrier containing such an organic substance may not be able to perform its intended function sufficiently. In such a case, a treatment for removing the organic substance is required by a baking treatment. Furthermore, depending on the type of the substrate, there are some that cannot be baked, and there is a problem that the type of the substrate is unavoidable.
As a method for producing a metal nanocolloid solution that does not use a protective colloid-forming agent, for example, a method is known in which a reducing agent is added to a solution in which a metal chloride is dissolved, and metal ions are reduced to generate metal fine particles. (For example, refer to Patent Document 4 and Non-Patent Document 1).
しかしながら、このような保護コロイド形成剤を含まない金属ナノコロイド液を用いて、コロイド粒子を基体に担持させる場合、自然吸着担持法が一般的に用いられるが、この方法では担持速度が遅く、担持濃度もあまり上げることができないという問題がある。
また、金属ナノコロイド液を用いて、金属担時体を製造する場合、1回の担時操作で、基体にできるだけ多量の金属ナノコロイド粒子を担持させることが作業上好ましく、したがって、金属ナノコロイド液におけるコロイド粒子の濃度を高めることが要求される。
しかしながら、前記の保護コロイド形成剤を用いない金属ナノコロイド液の製造方法においては、高濃度の金属ナノコロイド粒子を含む金属ナノコロイド液を調製しようとすると、コロイド粒子の凝集沈殿が容易に生じるという問題がある。これは、分散に寄与しているのが金属微粒子に吸着しているイオンのみなので、粒子間の距離が近くなりすぎると、静電遮蔽が起こり、反発力が不充分となって凝集が起こるものと推察される。
したがって、保護コロイド形成剤を含まず、かつ分散安定性の良好な高濃度の金属ナノコロイド液が求められている。なお、例えば金属として白金を用い、保護コロイド形成剤を用いない場合には、150質量ppm程度の濃度の金属ナノコロイド溶液を調製するのが限界であった。
However, when a colloidal particle is supported on a substrate using such a metal nanocolloid solution that does not contain a protective colloid-forming agent, a natural adsorption support method is generally used. There is a problem that the concentration cannot be increased too much.
In addition, when producing a metal carrier using a metal nanocolloid solution, it is preferable in terms of work to support as much metal nanocolloid particles as possible on a substrate in one operation. It is required to increase the concentration of colloidal particles in the liquid.
However, in the method for producing a metal nanocolloid liquid that does not use the protective colloid-forming agent, when an attempt is made to prepare a metal nanocolloid liquid containing a high concentration of metal nanocolloid particles, the colloidal particles are easily aggregated and precipitated. There's a problem. This is because only ions adsorbed to the metal fine particles contribute to the dispersion, and if the distance between the particles becomes too close, electrostatic shielding occurs and the repulsive force is insufficient, causing aggregation. It is guessed.
Therefore, there is a need for a high concentration metal nanocolloid solution that does not contain a protective colloid-forming agent and has good dispersion stability. For example, when platinum is used as the metal and the protective colloid forming agent is not used, it is a limit to prepare a metal nanocolloid solution having a concentration of about 150 ppm by mass.
本発明は、このような状況下でなされたもので、保護コロイド形成剤を実質上含まず、しかも金属ナノコロイド粒子を比較的高濃度で含有しても分散安定性が良好な金属ナノコロイド液を用い、金属ナノコロイド粒子を高速かつ高濃度に効率良く基体に担持させ、各種分野で有用な金属担持体を製造する方法、及びこの方法で得られた金属担持体を提供することを目的とするものである。 The present invention has been made under such circumstances, and it contains substantially no protective colloid-forming agent, and even if it contains metal nanocolloid particles at a relatively high concentration, the metal nanocolloid solution has good dispersion stability. And a method for producing a metal carrier useful in various fields, and a metal carrier obtained by this method. To do.
本発明者らは、前記目的を達成するために鋭意研究を重ねた結果、保護コロイド形成剤を含まず、好ましくは濃度が40〜2000質量ppm程度、より好ましくは120〜2000質量ppm、特に好ましくは500〜2000質量ppmの難凝集性金属ナノコロイド液を用い、電着法により金属ナノコロイド粒子を導電性基体に担持させることにより、その目的を達成し得ることを見出した。本発明は、かかる知見に基づいて完成したものである。
すなわち、本発明は、
(1)金属ナノコロイド液を用い、基体に金属ナノコロイド粒子を担持させて金属担持体を製造する方法において、基体として導電性基体を、金属ナノコロイド液として保護コロイド形成剤を実質上含まない金属ナノコロイド液を使用し、電着法により、前記基体に金属ナノコロイド粒子を担持させることを特徴とする金属担持体の製造方法、
(2)金属ナノコロイド液における保護コロイド形成剤の含有量が、金属ナノコロイド粒子に対し、全炭素として0〜200質量ppmである上記(1)に記載の金属担持体の製造方法、
(3)金属ナノコロイド液における金属ナノコロイド粒子の濃度が40〜2000質量ppmである上記(1)又は(2)に記載の金属担持体の製造方法、
(4)金属ナノコロイド液における金属ナノコロイド粒子の平均粒径が1〜200nmである上記(1)、(2)又は(3)に記載の金属担持体の製造方法、
(5)金属ナノコロイド液が、該コロイド液の製造過程で用いた還元剤を、金属ナノコロイド粒子を構成する金属の原子モル量に対し、0.03〜0.25倍の分子モル量含む上記(1)〜(4)のいずれかに記載の金属担持体の製造方法、
(6)金属ナノコロイド粒子が、白金、ルテニウム、パラジウム、ロジウム、レニウム、オスミウム及び金の中から選ばれる少なくとも一種の貴金属のナノコロイド粒子である上記(1)〜(5)のいずれかに記載の金属担持体の製造方法、
(7)導電性基体が、炭素系材料、導電性金属酸化物系材料又は金属系材料を素材とするものである上記(1)〜(6)のいずれかに記載の金属担持体の製造方法、
(8)導電性基体が、セラミックス系材料、非導電性金属酸化物系材料又は有機高分子系材料を素材とし、かつ表面に導電層を有するものである上記(1)〜(6)のいずれかに記載の金属担持体の製造方法、
(9)導電性基体が、金属ナノコロイド液の製造過程で用いた還元剤により、予め表面処理されたものである上記(1)〜(8)のいずれかに記載の金属担持体の製造方法、及び
(10)上記(1)〜(9)のいずれかに記載の製造方法により得られたことを特徴とする金属担持体、
を提供するものである。
As a result of intensive studies to achieve the above object, the inventors of the present invention do not include a protective colloid-forming agent, and preferably have a concentration of about 40 to 2000 ppm by mass, more preferably 120 to 2000 ppm by mass, and particularly preferably. Has found that the object can be achieved by supporting a metal nanocolloid particle on a conductive substrate by an electrodeposition method using a 500-2000 mass ppm hardly-aggregating metal nanocolloid solution. The present invention has been completed based on such findings.
That is, the present invention
(1) In a method for producing a metal carrier by supporting metal nanocolloid particles on a substrate using a metal nanocolloid solution, the substrate is substantially free of a conductive substrate and the metal nanocolloid solution as a protective colloid-forming agent. A method for producing a metal carrier, comprising using a metal nanocolloid solution, and carrying the metal nanocolloid particles on the substrate by electrodeposition;
(2) The method for producing a metal carrier according to (1), wherein the content of the protective colloid-forming agent in the metal nanocolloid liquid is 0 to 200 ppm by mass as the total carbon with respect to the metal nanocolloid particles,
(3) The method for producing a metal carrier according to (1) or (2) above, wherein the concentration of the metal nanocolloid particles in the metal nanocolloid liquid is 40 to 2000 ppm by mass,
(4) The method for producing a metal carrier according to (1), (2) or (3) above, wherein the average particle size of the metal nanocolloid particles in the metal nanocolloid liquid is 1 to 200 nm,
(5) The metal nanocolloid liquid contains 0.03 to 0.25 times the molecular molar amount of the reducing agent used in the production process of the colloidal liquid with respect to the atomic molar amount of the metal constituting the metal nanocolloid particles. The method for producing a metal carrier according to any one of (1) to (4) above,
(6) The metal nanocolloid particles according to any one of (1) to (5), wherein the metal nanocolloid particles are at least one kind of noble metal nanocolloid particles selected from platinum, ruthenium, palladium, rhodium, rhenium, osmium, and gold. A method for producing a metal carrier of
(7) The method for producing a metal carrier according to any one of (1) to (6), wherein the conductive substrate is made of a carbon-based material, a conductive metal oxide-based material, or a metal-based material. ,
(8) Any of the above (1) to (6), wherein the conductive substrate is made of a ceramic material, a nonconductive metal oxide material or an organic polymer material and has a conductive layer on the surface. A method for producing the metal carrier according to claim 1,
(9) The method for producing a metal carrier according to any one of (1) to (8), wherein the conductive substrate is surface-treated in advance with a reducing agent used in the production process of the metal nanocolloid liquid. And (10) a metal carrier obtained by the production method according to any one of (1) to (9) above,
Is to provide.
本発明によれば、保護コロイド形成剤を実質上含まず、しかも平均粒径1〜20nm程度の金属ナノコロイド粒子を、比較的高濃度で含有しても分散安定性が良好な金属ナノコロイド液を用い、電着法により金属ナノコロイド粒子を基体に高速かつ高濃度に効率よく担持させ、各種分野で有用な金属担持体を製造する方法、及びこの方法で得られた金属担持体を提供することができる。 According to the present invention, a metal nanocolloid liquid that is substantially free of a protective colloid-forming agent and has good dispersion stability even if it contains metal nanocolloid particles having an average particle diameter of about 1 to 20 nm at a relatively high concentration. A method for producing a metal carrier useful in various fields by efficiently supporting metal nanocolloid particles on a substrate at a high speed and a high concentration by an electrodeposition method, and a metal carrier obtained by this method is provided. be able to.
本発明の金属担持体の製造方法においては、基体として導電性基体を、金属ナノコロイド液として保護コロイド形成剤を実質上含まないコロイド液を使用し、電着法により、前記基体に金属ナノコロイド粒子を担持させることにより、金属担持体を製造する。
本発明で用いられる金属ナノコロイド液は、保護コロイド形成剤を実質上含まないものである。ここで、保護コロイド形成剤とは、従来コロイド粒子の分散安定性を保持するためにコロイド液に含有されているもので、コロイド粒子表面に付着して保護コロイドを形成する物質のことである。このような保護コロイド形成剤としては、例えばポリビニルアルコール、ポリビニルピロリドン、ゼラチンなどの水溶性高分子物質、界面活性剤、高分子キレート化剤(例えば、特開2000−279818号公報における〔0013〕に記載の化合物)などが挙げられる。
本発明においては、このような保護コロイド形成剤を実質上含まなくても、金属ナノコロイド粒子の分散安定性が良好であり、実用上十分な長期間、例えば3日〜30日間程度安定した分散性を保持する。なお、保護コロイド形成剤を実質上含まないとは、当該金属ナノコロイド液中の保護コロイド形成剤の含有量が、金属ナノコロイド粒子に対し、全炭素として0〜200質量ppm程度であることを意味する。
In the method for producing a metal carrier according to the present invention, a conductive substrate is used as the substrate, and a colloid solution substantially free of a protective colloid-forming agent is used as the metal nanocolloid solution. A metal carrier is produced by carrying the particles.
The metal nanocolloid liquid used in the present invention is substantially free of a protective colloid-forming agent. Here, the protective colloid-forming agent is a substance that is conventionally contained in a colloid liquid in order to maintain the dispersion stability of the colloidal particles, and adheres to the surface of the colloidal particles to form a protective colloid. Examples of such protective colloid-forming agents include water-soluble polymer substances such as polyvinyl alcohol, polyvinyl pyrrolidone, and gelatin, surfactants, and polymer chelating agents (for example, [0013] in JP-A-2000-279818). And the like).
In the present invention, even when such a protective colloid-forming agent is not substantially contained, the dispersion stability of the metal nanocolloid particles is good, and the dispersion is stable for a practically sufficient long period, for example, about 3 to 30 days. Retain sex. In addition, that the protective colloid forming agent is substantially not included means that the content of the protective colloid forming agent in the metal nanocolloid liquid is about 0 to 200 ppm by mass as the total carbon with respect to the metal nanocolloid particles. means.
当該金属ナノコロイド液においては、金属ナノコロイド粒子の濃度は、希薄濃度から高濃度まで広い範囲で選定することができるが、基体に金属ナノコロイド粒子を担持させる際の実用面及び該粒子の分散安定性の面から、通常40〜3000質量ppm、好ましくは250〜2000質量ppm、より好ましくは500〜2000質量ppm、特に好ましくは1000〜2000質量ppmの範囲である。
また、前記金属ナノコロイド粒子の平均粒径は、通常1〜20nm、好ましくは1〜10nmの範囲であるが、該金属ナノコロイド粒子を触媒に用いる場合には、触媒活性の点から、1.6〜5nmの範囲が好ましい。
金属ナノコロイド粒子の種類については特に制限はないが、白金、ルテニウム、パラジウム、ロジウム、レニウム、オスミウム及び金の中から選ばれる少なくとも一種の貴金属のナノコロイド粒子であることが好ましい。
In the metal nanocolloid liquid, the concentration of the metal nanocolloid particles can be selected in a wide range from a dilute concentration to a high concentration, but the practical aspect when the metal nanocolloid particles are supported on the substrate and the dispersion of the particles. From the viewpoint of stability, it is usually in the range of 40 to 3000 mass ppm, preferably 250 to 2000 mass ppm, more preferably 500 to 2000 mass ppm, and particularly preferably 1000 to 2000 mass ppm.
The average particle size of the metal nanocolloid particles is usually in the range of 1 to 20 nm, preferably 1 to 10 nm. When the metal nanocolloid particles are used as a catalyst, from the viewpoint of catalytic activity, 1. The range of 6-5 nm is preferable.
The type of metal nanocolloid particles is not particularly limited, but is preferably at least one noble metal nanocolloid particle selected from platinum, ruthenium, palladium, rhodium, rhenium, osmium, and gold.
本発明で用いる金属ナノコロイド液は、例えば以下のようにして製造することができる。
水としては、蒸留水、イオン交換水、限外ろ過水などの純水を十分に煮沸して、溶存酸素を除いたものを使用する。
上記の純水を用いて調製した金属塩水溶液に還元剤水溶液を、金属塩濃度が1×10-4〜15×10-4モル/リットル程度になるように、かつ還元剤が金属塩に対して、1〜20倍当量程度になるように加え、煮沸状態で30〜300分間程度反応を行ったのち、室温まで急冷して反応を停止させる。
次いで、所望に応じ、イオン交換樹脂を充填したカラムに、反応終了液を通し、未反応の金属塩及び還元剤を除去することで、希薄金属ナノコロイド液が得られる。除去の程度は、コロイド液の電気伝導度を測定することにより判断することができるが、100ミリリットルのコロイド液では、イオン交換樹脂がアンバーライトMB−1(商品名、オルガノ株式会社製)の場合、6ミリリットル程度で十分である。この際、イオン交換樹脂に吸着される金属ナノコロイド粒子は、極く僅かである。なお、後述する濃縮工程を行う場合は、濃縮後にイオン交換樹脂で処理することもできる。
The metal nanocolloid liquid used in the present invention can be produced, for example, as follows.
As the water, water obtained by sufficiently boiling pure water such as distilled water, ion-exchanged water, and ultrafiltered water and excluding dissolved oxygen is used.
The reducing agent aqueous solution is added to the metal salt aqueous solution prepared using the above pure water so that the metal salt concentration is about 1 × 10 −4 to 15 × 10 −4 mol / liter, and the reducing agent is added to the metal salt. In addition, the reaction is carried out in a boiling state for about 30 to 300 minutes, and then rapidly cooled to room temperature to stop the reaction.
Next, if necessary, the reaction-terminated liquid is passed through a column packed with an ion exchange resin to remove unreacted metal salt and reducing agent, thereby obtaining a diluted metal nanocolloid liquid. The degree of removal can be determined by measuring the electrical conductivity of the colloidal liquid. In the case of 100 ml of colloidal liquid, the ion exchange resin is Amberlite MB-1 (trade name, manufactured by Organo Corporation). About 6 milliliters is sufficient. At this time, very few metal nanocolloid particles are adsorbed on the ion exchange resin. In addition, when performing the concentration process mentioned later, it can also process with an ion exchange resin after concentration.
このようにして得られた希薄金属ナノコロイド液は、そのまま担持用として用いることができるが、高濃度の金属ナノコロイド液を担持用として用いる場合には、前記の希薄金属ナノコロイド液を、穏やかな条件(例えば非煮沸状態)にて加熱処理し、該コロイド液中の分散媒体を留去させ、濃縮することにより調製することができる。濃縮の条件によって、例えば煮沸状態になった場合などは、対流や生じる気泡の破裂などの影響によって、コロイド粒子の凝集が生じやすくなる。従って、コロイド粒子の凝集が生じないような穏やかな条件を選択することが好ましい。分散媒体が水である場合には、常圧下又は減圧下に50〜90℃程度の温度で、15分〜240分程度の時間をかけて水を留去させればよく、減圧度、温度及び濃縮の時間を変化させることによって、コロイド溶液の濃度を制御することができる。 The dilute metal nanocolloid solution thus obtained can be used as it is for supporting, but when a high concentration metal nanocolloid solution is used for supporting, the dilute metal nanocolloid solution is used as a gentle solution. It can be prepared by heat treatment under various conditions (for example, non-boiled state), distilling off the dispersion medium in the colloidal liquid, and concentrating. Depending on the concentration conditions, for example, when the liquid is boiled, colloidal particles are likely to aggregate due to the influence of convection or bursting of the generated bubbles. Therefore, it is preferable to select mild conditions that do not cause aggregation of colloidal particles. When the dispersion medium is water, water may be distilled off at a temperature of about 50 to 90 ° C. under normal pressure or reduced pressure over a period of about 15 to 240 minutes. By changing the time of concentration, the concentration of the colloidal solution can be controlled.
前記方法における還元剤としては、水に溶解するものであればとくに限定されるものではなく、アルコール類、クエン酸類、カルボン酸類、ケトン類、エーテル類、アルデヒド類またはエステル類が例示される。また、これらの二種以上を併用してもよい。アルコール類としては、メタノール、エタノール、1−プロパノール、2−プロパノール、エチレングリコール、グリセリンなどが例示される。クエン酸類としては、クエン酸や、クエン酸ナトリウム、クエン酸カリウム、クエン酸アンモニウムなどのクエン酸塩が例示される。カルボン酸類としては、ぎ酸、酢酸、フマル酸、リンゴ酸、コハク酸、アスパラギン酸及びそれらのカルボン酸塩などが例示される。ケトン類としては、アセトン、メチルエチルケトンなどが例示される。エーテル類としては、ジエチルエーテルなどが例示される。アルデヒド類としては、ホルムアルデヒド、アセトアルデヒドなどが例示される。エステル類としては、ぎ酸メチル、酢酸メチル、酢酸エチルなどが例示される。これらの中でも、還元性が高く、取り扱いも容易なクエン酸ナトリウムが特に好ましい。
平均粒径1〜20nm程度の安定な金属ナノコロイド粒子を生成する観点からは、還元剤としてアルコール類、クエン酸類またはカルボン酸類が好ましい。特に平均粒径1〜5nmの安定な金属のコロイド粒子を生成するためには、クエン酸類が好適である。ただし、該コロイド粒子は、触媒活性の面では、その平均粒径が1.6nm以上であることが好ましい。
The reducing agent in the method is not particularly limited as long as it is soluble in water, and examples thereof include alcohols, citric acids, carboxylic acids, ketones, ethers, aldehydes, and esters. Two or more of these may be used in combination. Examples of alcohols include methanol, ethanol, 1-propanol, 2-propanol, ethylene glycol, glycerin and the like. Examples of citric acids include citric acid and citrates such as sodium citrate, potassium citrate, and ammonium citrate. Examples of carboxylic acids include formic acid, acetic acid, fumaric acid, malic acid, succinic acid, aspartic acid, and carboxylates thereof. Examples of ketones include acetone and methyl ethyl ketone. Examples of ethers include diethyl ether. Examples of aldehydes include formaldehyde and acetaldehyde. Examples of the esters include methyl formate, methyl acetate, and ethyl acetate. Among these, sodium citrate is particularly preferable because of its high reducibility and easy handling.
From the viewpoint of producing stable metal nanocolloid particles having an average particle size of about 1 to 20 nm, alcohols, citric acids or carboxylic acids are preferred as the reducing agent. In particular, citric acids are suitable for producing stable metal colloidal particles having an average particle diameter of 1 to 5 nm. However, the colloidal particles preferably have an average particle size of 1.6 nm or more in terms of catalytic activity.
また、反応媒体としては、例えば水、アルコール類、ケトン類、エステル類、エーテル類などを用いることができる。アルコール類としては、メタノール、エタノール、1−プロパノール、2−プロパノールなどが例示でき、ケトン類としては、アセトン、メチルエチルケトン、メチルイソブチルケトンなどが例示できる。また、エステル類としては、ぎ酸メチル、酢酸メチル、酢酸エチルなどが、エーテル類としては、メチルエチルエーテル、ジエチルエーテルなどが例示できる。前記反応媒体は一種を単独で用いてもよく、二種以上を組み合わせて用いても良いが、水又はアルコール類あるいはそれらの混合物のような水性媒体が好ましい。 As the reaction medium, for example, water, alcohols, ketones, esters, ethers and the like can be used. Examples of alcohols include methanol, ethanol, 1-propanol, and 2-propanol. Examples of ketones include acetone, methyl ethyl ketone, and methyl isobutyl ketone. Examples of the esters include methyl formate, methyl acetate, and ethyl acetate. Examples of the ethers include methyl ethyl ether and diethyl ether. The reaction medium may be used singly or in combination of two or more, but an aqueous medium such as water or alcohols or a mixture thereof is preferable.
一方、金属塩は、反応媒体に溶解し、還元剤により還元されるもので、コロイド粒子となり得るものであれば、その種類はとくに限定されるものではない。例えば、白金、ルテニウム、パラジウム、ロジウム、レニウム、オスミウム、金、鉛、イリジウム、コバルト、鉄、ニッケル、銅、スズなど、好ましくは白金、ルテニウム、パラジウム、ロジウム、レニウム、オスミウム、金などの貴金属の塩化物、硝酸塩、硫酸塩またはこれらの金属錆化合物などが挙げられる。また、これらの二種以上を併用してもよい。二種の金属塩を併用する場合は、合金のコロイド粒子を製造することができる。金属塩として白金塩を用いた場合は、コロイド粒子の粒径が特に小さくなり、平均粒径1〜5nm程度の安定なコロイド粒子が得られる。とくに、塩化白金酸を使用すれば、コロイド粒子の粒径の均一化を一層図ることができる。 On the other hand, the type of metal salt is not particularly limited as long as it is dissolved in the reaction medium and reduced by the reducing agent and can be colloidal particles. For example, platinum, ruthenium, palladium, rhodium, rhenium, osmium, gold, lead, iridium, cobalt, iron, nickel, copper, tin, etc., preferably platinum, ruthenium, palladium, rhodium, rhenium, osmium, gold, etc. Examples include chlorides, nitrates, sulfates, and metal rust compounds thereof. Two or more of these may be used in combination. When two kinds of metal salts are used in combination, alloy colloidal particles can be produced. When a platinum salt is used as the metal salt, the particle size of the colloidal particles is particularly small, and stable colloidal particles having an average particle size of about 1 to 5 nm can be obtained. In particular, if chloroplatinic acid is used, the particle size of the colloidal particles can be made more uniform.
本発明においては、前記金属ナノコロイド液を用いて、金属ナノコロイド粒子を基体に担持させる方法として、電着法が採用されるため、基体として導電性基体が用いられる。
この導電性基体としては、例えば(1)炭素系材料、導電性金属酸化物系材料、金属系材料を素材とするもの、(2)セラミックス系材料、非導電性金属酸化物系材料、有機高分子系材料などを素材とし、かつ少なくともその表面に導電層を有するものを用いることができる。
前記(1)の炭素系材料としては、活性炭、木炭、カーボンブラック、グラファイト、炭素繊維などが例示され、導電性金属酸化物系材料としては、代表的なものとして錫ドープ酸化インジウム(ITO)などが例示される。金属系材料としては鋳鉄、鋼、鉄系合金、アルミニウム及びその合金、マグネシウム及びその合金、亜鉛及びその合金、銅及びその合金、チタン及びその合金、ニッケル、コバルト及びそれらの合金などが例示される。
In the present invention, since the electrodeposition method is employed as a method for supporting the metal nanocolloid particles on the substrate using the metal nanocolloid solution, a conductive substrate is used as the substrate.
Examples of the conductive substrate include (1) carbon-based material, conductive metal oxide-based material, metal-based material, (2) ceramic-based material, non-conductive metal oxide-based material, organic high A material using a molecular material or the like and having a conductive layer on at least the surface thereof can be used.
Examples of the carbon-based material (1) include activated carbon, charcoal, carbon black, graphite, and carbon fiber. Typical examples of the conductive metal oxide-based material include tin-doped indium oxide (ITO). Is exemplified. Examples of metal materials include cast iron, steel, iron alloys, aluminum and alloys thereof, magnesium and alloys thereof, zinc and alloys thereof, copper and alloys thereof, titanium and alloys thereof, nickel, cobalt, and alloys thereof. .
一方、前記(2)のセラミックス系材料や非導電性金属酸化物系材料としては、アルミナ、チタニア、マグネシア、シリカ、シリカ・アルミナ、ジルコニア、ゼオライト、ガラスなどが例示される。また、有機高分子系材料としては、ポリエチレンテレフタレート、ポリブチレンテレフタレート、ポリエチレンナフタレート、ポリカーボネートなどのポリエステル系樹脂、ポリメチルメタクリレートなどのアクリル系樹脂、ポリエチレン、ポリプロピレン、ポリメチルペンテン、脂環式構造含有重合体などのポリオレフィン系樹脂、セロファン、ジアセチルセルロース、トリアセチルセルロース、アセチルセルロースブチレートなどのセルロース類、ポリ塩化ビニル、ポリ塩化ビニリデン、ポリビニルアルコール、エチレン−酢酸ビニル共重合体、ポリフェニレンエーテル、ポリフェニレンサルファイド、ポリスチレン、ポリスルホン、ポリエーテルエーテルケトン、ポリエーテルスルホン、ポリエーテルイミド、ポリイミド、フッ素樹脂、ポリアミドなどが例示される。これらの材料は非導電性であるので、少なくともその表面に導電性を付与することが必要である。すなわち、表面に導電層を形成するか、又は導電性微粒子や導電性繊維を原料に混合して形成したものを用いる。導電層を形成する材料又は混合される導電性微粒子や導電性繊維などの材料は、導電性を有するものであればよく、特に制限はない。 On the other hand, examples of the ceramic material and non-conductive metal oxide material (2) include alumina, titania, magnesia, silica, silica / alumina, zirconia, zeolite, and glass. Organic polymer materials include polyethylene terephthalate, polybutylene terephthalate, polyethylene naphthalate, polyester resins such as polycarbonate, acrylic resins such as polymethyl methacrylate, polyethylene, polypropylene, polymethylpentene, and alicyclic structures. Polyolefin resins such as polymers, celluloses such as cellophane, diacetylcellulose, triacetylcellulose, acetylcellulose butyrate, polyvinyl chloride, polyvinylidene chloride, polyvinyl alcohol, ethylene-vinyl acetate copolymer, polyphenylene ether, polyphenylene sulfide , Polystyrene, polysulfone, polyetheretherketone, polyethersulfone, polyetherimide, polyimide, fluororesin Polyamide are exemplified. Since these materials are non-conductive, it is necessary to impart conductivity to at least the surface thereof. That is, a conductive layer is formed on the surface, or a conductive fine particle or conductive fiber mixed with a raw material is used. The material for forming the conductive layer or the mixed material such as conductive fine particles and conductive fibers is not particularly limited as long as it has conductivity.
前記導電性基体の形態、形状については特に制限はなく、棒状体、繊維、織布、不織布、フィルム、シート、板状体などが例示される。
本発明においては、前述の金属ナノコロイド液を用い、電着法により前記導電性基体に金属ナノコロイド粒子を担持させる。この電着法としては、例えば、従来水性塗料を用いた電着塗装(電気泳動塗装ともいわれる。)において慣用されている方法を採用することができる。
具体的には、導電性基体に、金属ナノコロイド粒子の荷電とは逆符号の直流電圧を印加して作用電極とする。この際、対極としては、金属ナノコロイド液槽を鋼板などで作製し、そのまま該槽を対極としてよく、あるいは上記槽を絶縁材料で作製し、別途対極を設けてもよい。
There is no restriction | limiting in particular about the form of the said electroconductive base | substrate, A rod-shaped body, a fiber, a woven fabric, a nonwoven fabric, a film, a sheet | seat, a plate-shaped body etc. are illustrated.
In the present invention, metal nanocolloid particles are supported on the conductive substrate by the electrodeposition method using the metal nanocolloid solution described above. As this electrodeposition method, for example, a method conventionally used in electrodeposition coating (also referred to as electrophoretic coating) using a water-based paint can be employed.
Specifically, a DC voltage having a sign opposite to the charge of the metal nanocolloid particles is applied to the conductive substrate to form a working electrode. In this case, as the counter electrode, the metal nanocolloid liquid tank may be made of a steel plate and the tank may be used as the counter electrode as it is, or the tank may be made of an insulating material and a separate counter electrode may be provided.
このようにして、導電性基体からなる作用電極と対極との間に直流電流を通電すると、金属ナノコロイド液中のコロイド粒子は、導電性基体に集まり、ここで電荷を失って該基体表面に定着することにより担持される。この際、直流電源としては、例えばシリコン整流器を用いて交流を直流に変換して用いることができる。
この電着法においては、金属ナノコロイド粒子の分散安定性をさらに向上させるために、使用する金属ナノコロイド液の製造過程で用いた還元剤を、金属ナノコロイド粒子を構成する金属のモル量に対し、0.03〜0.25倍モル程度の割合で、該金属ナノコロイド液に含有させることが望ましい。これにより、例えばニッケルイオンなどの共存イオンに起因する金属ナノコロイド粒子の凝集を抑制することができる。前記還元剤としては、例えばクエン酸ナトリウムが好適である。
In this way, when a direct current is passed between the working electrode made of a conductive substrate and the counter electrode, the colloidal particles in the metal nanocolloid liquid gather on the conductive substrate, where they lose their charge and are applied to the surface of the substrate. It is carried by fixing. At this time, as the DC power source, for example, a silicon rectifier can be used by converting AC to DC.
In this electrodeposition method, in order to further improve the dispersion stability of the metal nanocolloid particles, the reducing agent used in the production process of the metal nanocolloid liquid used is changed to the molar amount of the metal constituting the metal nanocolloid particles. On the other hand, the metal nanocolloid liquid is desirably contained in a ratio of about 0.03 to 0.25 times mole. Thereby, aggregation of the metal nano colloid particle resulting from coexisting ions, such as nickel ion, can be suppressed, for example. As the reducing agent, for example, sodium citrate is suitable.
また、この電着法による金属ナノコロイド粒子の担持においては、コロイド粒子の荷電状態と基体表面の荷電状態の組合わせにより、担持されにくい組合わせが存在することがある。この場合には、該基体を、使用する金属ナノコロイド液の製造過程で用いた還元剤、好ましくはクエン酸ナトリウムにより、予め表面処理することが望ましい。
このような本発明の方法により、電着法による金属ナノコロイド粒子を担持させる場合、下記の効果を奏する。
(1)金属ナノコロイド粒子の特性を維持したまま担持することができる。
(2)保護コロイド形成剤を含有しないコロイド液を使用するにもかかわらず、担持速度が速く、高濃度に担持することができる。
(3)共存イオンの存在下でも、保護コロイド形成剤なしで、コロイド粒子の安定分散を維持することができる。
(4)電荷的に担持されにくい基体に対しても担持を促進することができる。
本発明はまた、前述の電着法により、導電性基体に金属ナノコロイド粒子を担持させることにより得られた金属担持体をも提供する。
In addition, in the loading of metal nanocolloid particles by this electrodeposition method, there may be a combination that is difficult to carry depending on the combination of the charged state of the colloidal particles and the charged state of the substrate surface. In this case, it is desirable that the substrate is surface-treated in advance with a reducing agent, preferably sodium citrate, used in the production process of the metal nanocolloid liquid to be used.
When the metal nano-colloid particles by the electrodeposition method are supported by the method of the present invention, the following effects are obtained.
(1) It can be supported while maintaining the properties of metal nanocolloid particles.
(2) Despite the use of a colloid liquid that does not contain a protective colloid-forming agent, the loading speed is high and the film can be loaded at a high concentration.
(3) Even in the presence of coexisting ions, stable dispersion of colloidal particles can be maintained without a protective colloid-forming agent.
(4) Supporting can be promoted even on a substrate that is not easily supported by charge.
The present invention also provides a metal carrier obtained by supporting metal nanocolloid particles on a conductive substrate by the electrodeposition method described above.
次に、本発明を実施例により、さらに詳細に説明するが、本発明は、この例によってなんら限定されるものではない。
実施例1
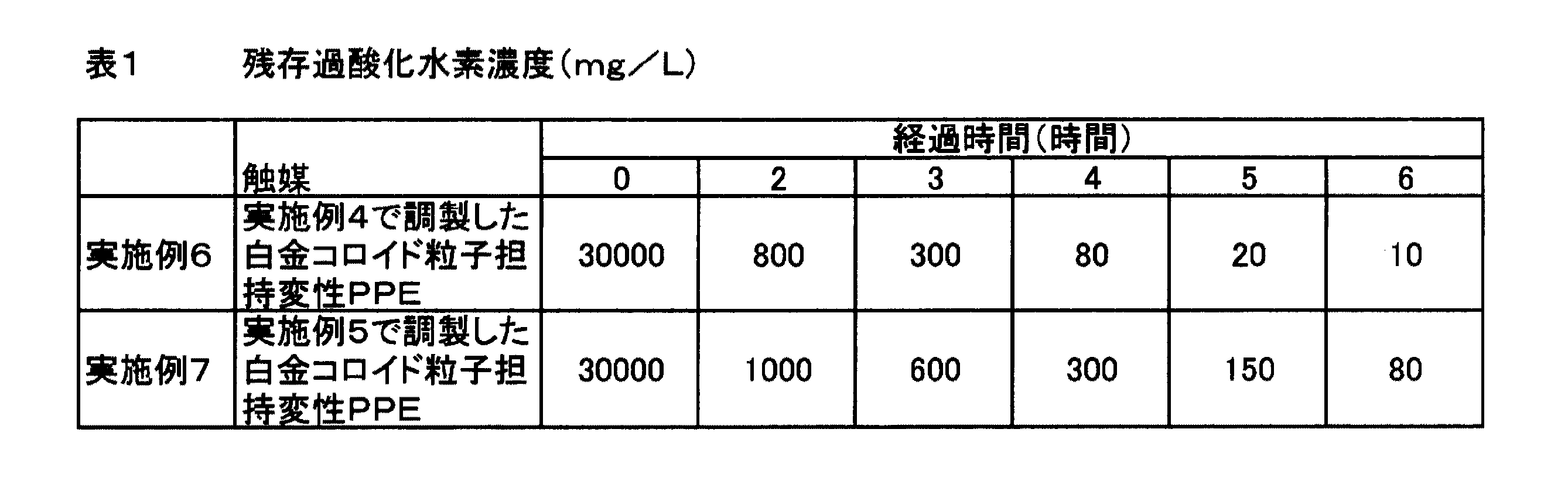
1500mlフラスコ、100ml三角フラスコ、200ml三角フラスコ、還流コンデンサー及び撹拌子を王水中に一昼夜浸し、イオン交換及び限外濾過した純水を用いて前記器具を十分に洗浄した。イオン交換及び限外濾過した純水850mlと撹拌子とを前記1500mlフラスコに投入し、還流コンデンサーをフラスコ上部に設置して、これを100℃まで加熱・昇温した。純水中の溶存酸素を取り除くため、そのまま1時間煮沸を行った。一方、100ml三角フラスコに、塩化白金酸(H2PtCl6・6H2O)400mg(白金として150mg)を秤量して投入し、そこにイオン交換及び限外濾過した純水を加えて50mlとした。また、200ml三角フラスコに、クエン酸ナトリウム1.0gを秤量して投入し、イオン交換及び限外濾過した純水を加えて100mlとした。純水の溶存酸素を取り除いた後、1500mlフラスコに100ml三角フラスコからテトラクロロ白金酸水溶液を投入し、再度100℃まで加熱・昇温した。さらに、溶存酸素を除去するため、30分間煮沸を行った。続いて、200mlフラスコからクエン酸ナトリウム水溶液を煮沸状態が維持されるように徐々に添加した。この反応溶液において、白金濃度は150mg/L=7.7×10-4mol/L=3.08×10-3Nであり、白金のモル濃度に対するクエン酸ナトリウムのモル濃度比は13.2となる。また、クエン酸ナトリウムは一電子供与体として機能するから、白金の当量濃度に対するクエン酸ナトリウムの当量濃度の比は3.3となる。
EXAMPLES Next, although an Example demonstrates this invention further in detail, this invention is not limited at all by this example.
Example 1
A 1500 ml flask, a 100 ml Erlenmeyer flask, a 200 ml Erlenmeyer flask, a reflux condenser and a stirring bar were immersed in aquatic water all day and night, and the instrument was thoroughly washed with ion-exchanged and ultrafiltered pure water. 850 ml of ion-exchanged and ultrafiltered pure water and a stirrer were put into the 1500 ml flask, a reflux condenser was installed at the top of the flask, and this was heated to 100 ° C. and heated. In order to remove dissolved oxygen in pure water, boiling was performed for 1 hour. On the other hand, 400 mg (150 mg as platinum) of chloroplatinic acid (H 2 PtCl 6 .6H 2 O) was weighed into a 100 ml Erlenmeyer flask, and ion-exchanged and ultrafiltered pure water was added thereto to make 50 ml. . Further, 1.0 g of sodium citrate was weighed and put into a 200 ml Erlenmeyer flask, and ion exchanged and ultrafiltered pure water was added to make 100 ml. After removing dissolved oxygen from pure water, an aqueous tetrachloroplatinic acid solution was added to a 1500 ml flask from a 100 ml Erlenmeyer flask, and the mixture was heated to 100 ° C. and heated again. Furthermore, in order to remove dissolved oxygen, boiling was performed for 30 minutes. Subsequently, an aqueous sodium citrate solution was gradually added from a 200 ml flask so as to maintain the boiling state. In this reaction solution, the platinum concentration was 150 mg / L = 7.7 × 10 −4 mol / L = 3.08 × 10 −3 N, and the molar concentration ratio of sodium citrate to the molar concentration of platinum was 13.2. It becomes. Further, since sodium citrate functions as a one-electron donor, the ratio of the equivalent concentration of sodium citrate to the equivalent concentration of platinum is 3.3.
クエン酸ナトリウム水溶液を1500mlフラスコに全て添加した後、煮沸状態で還元反応を継続させ、反応開始から90分後に反応を止めて、その反応液を室温まで急冷した。冷却した反応溶液をイオン交換樹脂アンバーライトMB-1(オルガノ株式会社製)を詰めたカラムに通し、反応溶液中に残存する金属イオンおよび還元剤を取り除いて安定な白金コロイド液を得た。この白金コロイド液について、プラズマ発光分光分析法により白金コロイド粒子の濃度を測定すると共に、透過型電子顕微鏡を用いて、その平均粒径を測定した。その結果、白金コロイド粒子の濃度は120mg/L、平均粒径は1.1nmであった。
次に、上記白金コロイド液中に、基体である導電膜付きガラスを浸漬し、下記の条件で直流電圧を印加することによって、下記に示す電圧印加時間のいずれにおいても、白金コロイド粒子をガラス表面に担持させることができた。
<電圧印加条件>
基体 : ITO膜付きガラス(作用極)
対極 : 白金線
電圧 : 0〜3Vの範囲で掃引(掃引速度:0.5V/分)
電圧印加時間 : 10分、60分、300分
電着温度 : 25℃(室温)
また、上記白金コロイド液25mlにクエン酸ナトリウムを添加しないもの、及びクエン酸ナトリウムを15.0ミリモル/L濃度になるように添加したものに、それぞれ1.7ミリモルのニッケルイオンを添加したところ、クエン酸ナトリウムを添加しないコロイド液は、凝集・沈殿が確認されたが、クエン酸ナトリウムを添加したコロイド液では、凝集・沈殿は発生しなかった。
After all the sodium citrate aqueous solution was added to the 1500 ml flask, the reduction reaction was continued in a boiling state, the reaction was stopped 90 minutes after the start of the reaction, and the reaction solution was rapidly cooled to room temperature. The cooled reaction solution was passed through a column packed with ion-exchange resin Amberlite MB-1 (manufactured by Organo Corporation), and metal ions and reducing agent remaining in the reaction solution were removed to obtain a stable platinum colloid solution. About this platinum colloid liquid, while measuring the density | concentration of the platinum colloid particle by the plasma emission spectrometry, the average particle diameter was measured using the transmission electron microscope. As a result, the concentration of the colloidal platinum particles was 120 mg / L, and the average particle size was 1.1 nm.
Next, by immersing the glass with a conductive film as a substrate in the platinum colloid liquid and applying a DC voltage under the following conditions, the platinum colloid particles are attached to the glass surface at any of the voltage application times shown below. It was possible to make it supported on.
<Voltage application conditions>
Base: Glass with ITO film (working electrode)
Counter electrode: Platinum wire Voltage: Sweep in the range of 0-3V (sweep speed: 0.5V / min)
Voltage application time: 10 minutes, 60 minutes, 300 minutes Electrodeposition temperature: 25 ° C. (room temperature)
Moreover, when adding 1.7 mmol of nickel ions to the platinum colloid solution 25 ml without adding sodium citrate and adding sodium citrate to a concentration of 15.0 mmol / L, Aggregation / precipitation was confirmed in the colloidal solution to which sodium citrate was not added, but aggregation / precipitation did not occur in the colloidal solution to which sodium citrate was added.
実施例2
実施例1で調製した白金コロイド液を濃縮し、白金コロイド粒子の濃度1000mg/Lの白金コロイド液を調製した。実施例1と同様に評価した結果、上記に示す電圧印加時間のいずれにおいても、白金コロイド粒子をガラス表面に担持させることができた。
Example 2
The platinum colloid solution prepared in Example 1 was concentrated to prepare a platinum colloid solution having a platinum colloid particle concentration of 1000 mg / L. As a result of evaluation in the same manner as in Example 1, the colloidal platinum particles could be supported on the glass surface at any of the voltage application times shown above.
実施例3
塩化白金酸(H2PtCl6・6H2O)450mgを原料として用いたこと以外は実施例1と同様にして安定な白金コロイド液を得た。実施例1と同様に評価したところ、白金コロイド粒子の濃度は135mg/L、平均粒径は1.1nmであった。
無電解めっきの方法で表面にNi導電性層を設けた変性PPE(非結晶質ポリフェニレンエーテル)からなる基体を上記白金コロイド液中に浸漬し、下記の条件で直流電圧を印加することによって、白金コロイド粒子を変性PPE基体表面に担持させることができた。
<電圧印加条件>
基体 : 変性PPE(Ni導電性層あり、大きさ10mm×30mm、厚み2mm)
対極 : ステンレス鋼(SUS304)
電圧 : 5V
電圧印加時間 : 10分
電着温度 : 25℃(室温)
Example 3
A stable platinum colloidal solution was obtained in the same manner as in Example 1 except that 450 mg of chloroplatinic acid (H 2 PtCl 6 .6H 2 O) was used as a raw material. When evaluated in the same manner as in Example 1, the concentration of the colloidal platinum particles was 135 mg / L, and the average particle size was 1.1 nm.
By immersing a substrate made of modified PPE (amorphous polyphenylene ether) having a Ni conductive layer on the surface by an electroless plating method in the above-mentioned platinum colloid solution and applying a DC voltage under the following conditions, Colloidal particles could be supported on the modified PPE substrate surface.
<Voltage application conditions>
Substrate: Modified PPE (with Ni conductive layer, size 10 mm x 30 mm, thickness 2 mm)
Counter electrode: Stainless steel (SUS304)
Voltage: 5V
Voltage application time: 10 minutes Electrodeposition temperature: 25 ° C (room temperature)
実施例4
無電解めっきの方法で表面にAg導電性層を設けた変性PPEからなる基体を、実施例3で調製した白金コロイド液中に浸漬し、下記の条件で直流電圧を印加することによって、白金コロイド粒子を変性PPE基体表面に担持させることができた。
<電圧印加条件>
基体 : 変性PPE(Ag導電性層あり、大きさ10mm×30mm、厚み2mm)
対極 : ステンレス鋼(SUS304)
電圧 : 8V
電圧印加時間 : 15分
電着温度 : 25℃(室温)
Example 4
A substrate made of modified PPE having an Ag conductive layer provided on the surface thereof by an electroless plating method is immersed in the platinum colloid solution prepared in Example 3, and a DC voltage is applied under the following conditions to thereby form a platinum colloid. The particles could be supported on the modified PPE substrate surface.
<Voltage application conditions>
Substrate: Modified PPE (with Ag conductive layer, size 10 mm x 30 mm, thickness 2 mm)
Counter electrode: Stainless steel (SUS304)
Voltage: 8V
Voltage application time: 15 minutes Electrodeposition temperature: 25 ° C (room temperature)
実施例5
電圧印加時間を8分としたこと以外は実施例4と同様に行ったところ、同様に白金コロイド粒子を変性PPE基体表面に担持させることができた。
Example 5
When the same procedure as in Example 4 was performed except that the voltage application time was 8 minutes, platinum colloidal particles could be similarly supported on the modified PPE substrate surface.
実施例6及び7
実施例4及び実施例5で得られた白金コロイド担持変性PPEを試料として、濃度3質量%の過酸化水素に浸漬し、その後の残留過酸化水素の濃度を測定することにより、過酸化水素の分解速度を評価した。その結果、経過時間(単位:時間)と残存濃度(単位:mg/L)について表1の結果を得た。この結果から、電圧印加条件の選択により貴金属コロイド担持量の制御が可能であること、すなわち、電圧印加条件の選択により所望の触媒活性を有する触媒を製造することができることが明らかとなった。よって、消毒、殺菌及び漂白などの用途においては、消毒などの作用と分解速度の調節が容易である。
Examples 6 and 7
The platinum colloid-supported modified PPE obtained in Example 4 and Example 5 was used as a sample, immersed in hydrogen peroxide having a concentration of 3% by mass, and then the concentration of residual hydrogen peroxide was measured. The degradation rate was evaluated. As a result, the results in Table 1 were obtained for the elapsed time (unit: time) and the residual concentration (unit: mg / L). From this result, it became clear that the amount of the precious metal colloid supported can be controlled by selecting the voltage application condition, that is, a catalyst having a desired catalytic activity can be produced by selecting the voltage application condition. Therefore, in applications such as disinfection, sterilization, and bleaching, it is easy to adjust the action of disinfection and the degradation rate.
本発明の金属担持体の製造方法によれば、保護コロイド形成剤を実質上含まず、しかも平均粒径1〜20nm程度の金属ナノコロイド粒子を、比較的高濃度で含有しても分散安定性が良好な金属ナノコロイド液を用い、電着法により金属ナノコロイド粒子を基体に高速かつ高濃度に効率よく担持させることができる。
本発明の方法は、例えば(1)導電性粒子、(2)消毒薬等の分解触媒、(3)自動車排気ガスの浄化用触媒、(4)光触媒薄膜、(5)燃料電池用の改質触媒、シフト反応触媒、電極触媒などの製造に好適に用いられる。
According to the method for producing a metal carrier of the present invention, even if a metal colloidal particle having an average particle diameter of about 1 to 20 nm is contained at a relatively high concentration, the dispersion stability is substantially eliminated. Can be efficiently supported at high speed and at a high concentration on a substrate by electrodeposition.
The method of the present invention includes, for example, (1) conductive particles, (2) a decomposition catalyst such as a disinfectant, (3) a catalyst for purifying automobile exhaust gas, (4) a photocatalytic thin film, and (5) reforming for a fuel cell. It is suitably used for the production of a catalyst, shift reaction catalyst, electrode catalyst and the like.
Claims (10)
A metal carrier obtained by the production method according to claim 1.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004362711A JP2005199267A (en) | 2003-12-15 | 2004-12-15 | Metal carrier and method for manufacturing the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003416148 | 2003-12-15 | ||
| JP2004362711A JP2005199267A (en) | 2003-12-15 | 2004-12-15 | Metal carrier and method for manufacturing the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2005199267A true JP2005199267A (en) | 2005-07-28 |
Family
ID=34829122
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2004362711A Withdrawn JP2005199267A (en) | 2003-12-15 | 2004-12-15 | Metal carrier and method for manufacturing the same |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP2005199267A (en) |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006051825A1 (en) * | 2004-11-09 | 2006-05-18 | Nippon Sheet Glass Company, Limited | Method for producing noble metal-loaded body |
| WO2007081054A1 (en) * | 2006-01-12 | 2007-07-19 | Kurita Water Industries Ltd. | Method and apparatus for removing hydrogen peroxide |
| JP2008104905A (en) * | 2006-10-23 | 2008-05-08 | Catalysts & Chem Ind Co Ltd | Method for manufacturing catalyst |
| JP2009228067A (en) * | 2008-03-24 | 2009-10-08 | Tanaka Kikinzoku Kogyo Kk | Colloidal solution of platinum and production process therefor |
| JP2012196654A (en) * | 2011-03-23 | 2012-10-18 | Toyota Central R&D Labs Inc | Catalyst for decomposition and removal of ozone, method of producing the same, and ozone decomposition/removal method using the same |
| KR101729885B1 (en) * | 2013-06-25 | 2017-04-24 | 가켄 테크 가부시키가이샤 | Flake-like silver powder, conductive paste, and method for producing flake-like silver powder |
| JP2017110296A (en) * | 2015-12-14 | 2017-06-22 | ローム アンド ハース エレクトロニック マテリアルズ エルエルシーRohm and Haas Electronic Materials LLC | Environmentally friendly stable catalyst for electroless metalization of printed circuit board and through-hole |
| JP2017110297A (en) * | 2015-12-14 | 2017-06-22 | ローム アンド ハース エレクトロニック マテリアルズ エルエルシーRohm and Haas Electronic Materials LLC | Environmentally friendly stable catalyst for electroless metalization of printed circuit board and through-hole |
| JP2017110298A (en) * | 2015-12-14 | 2017-06-22 | ローム アンド ハース エレクトロニック マテリアルズ エルエルシーRohm and Haas Electronic Materials LLC | Environmentally friendly stable catalyst for electroless metalization of printed circuit board and through-hole |
| JP2018501644A (en) * | 2014-11-18 | 2018-01-18 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | Porous interconnected corrugated carbon based network (ICCN) composites |
| JP2018111885A (en) * | 2005-08-03 | 2018-07-19 | ユニバシダデ デ サンチャゴ デ コンポステラ | Atomic quantum clusters, production method thereof and methods of use thereof |
| US10655020B2 (en) | 2015-12-22 | 2020-05-19 | The Regents Of The University Of California | Cellular graphene films |
| US10847852B2 (en) | 2014-06-16 | 2020-11-24 | The Regents Of The University Of California | Hybrid electrochemical cell |
| US10892109B2 (en) | 2016-01-22 | 2021-01-12 | The Regents Of The University Of California | High-voltage devices |
| US10938021B2 (en) | 2016-08-31 | 2021-03-02 | The Regents Of The University Of California | Devices comprising carbon-based material and fabrication thereof |
| US10938032B1 (en) | 2019-09-27 | 2021-03-02 | The Regents Of The University Of California | Composite graphene energy storage methods, devices, and systems |
| US11004618B2 (en) | 2012-03-05 | 2021-05-11 | The Regents Of The University Of California | Capacitor with electrodes made of an interconnected corrugated carbon-based network |
| US11062855B2 (en) | 2016-03-23 | 2021-07-13 | The Regents Of The University Of California | Devices and methods for high voltage and solar applications |
| US11097951B2 (en) | 2016-06-24 | 2021-08-24 | The Regents Of The University Of California | Production of carbon-based oxide and reduced carbon-based oxide on a large scale |
| US11133134B2 (en) | 2017-07-14 | 2021-09-28 | The Regents Of The University Of California | Simple route to highly conductive porous graphene from carbon nanodots for supercapacitor applications |
| US11397173B2 (en) | 2011-12-21 | 2022-07-26 | The Regents Of The University Of California | Interconnected corrugated carbon-based network |
-
2004
- 2004-12-15 JP JP2004362711A patent/JP2005199267A/en not_active Withdrawn
Cited By (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006051825A1 (en) * | 2004-11-09 | 2006-05-18 | Nippon Sheet Glass Company, Limited | Method for producing noble metal-loaded body |
| JP7190818B2 (en) | 2005-08-03 | 2022-12-16 | ユニバシダデ デ サンチャゴ デ コンポステラ | Atomic Quantum Clusters, Methods of Making Them and Methods of Using Them |
| JP2018111885A (en) * | 2005-08-03 | 2018-07-19 | ユニバシダデ デ サンチャゴ デ コンポステラ | Atomic quantum clusters, production method thereof and methods of use thereof |
| WO2007081054A1 (en) * | 2006-01-12 | 2007-07-19 | Kurita Water Industries Ltd. | Method and apparatus for removing hydrogen peroxide |
| JP2007185587A (en) * | 2006-01-12 | 2007-07-26 | Kurita Water Ind Ltd | Method and device for removing hydrogen peroxide |
| JP2008104905A (en) * | 2006-10-23 | 2008-05-08 | Catalysts & Chem Ind Co Ltd | Method for manufacturing catalyst |
| JP2009228067A (en) * | 2008-03-24 | 2009-10-08 | Tanaka Kikinzoku Kogyo Kk | Colloidal solution of platinum and production process therefor |
| JP2012196654A (en) * | 2011-03-23 | 2012-10-18 | Toyota Central R&D Labs Inc | Catalyst for decomposition and removal of ozone, method of producing the same, and ozone decomposition/removal method using the same |
| US11397173B2 (en) | 2011-12-21 | 2022-07-26 | The Regents Of The University Of California | Interconnected corrugated carbon-based network |
| US11257632B2 (en) | 2012-03-05 | 2022-02-22 | The Regents Of The University Of California | Capacitor with electrodes made of an interconnected corrugated carbon-based network |
| US11004618B2 (en) | 2012-03-05 | 2021-05-11 | The Regents Of The University Of California | Capacitor with electrodes made of an interconnected corrugated carbon-based network |
| US11915870B2 (en) | 2012-03-05 | 2024-02-27 | The Regents Of The University Of California | Capacitor with electrodes made of an interconnected corrugated carbon-based network |
| US10071420B2 (en) | 2013-06-25 | 2018-09-11 | Kaken Tech Co., Ltd. | Flake-like silver powder, conductive paste, and method for producing flake-like silver powder |
| KR101729885B1 (en) * | 2013-06-25 | 2017-04-24 | 가켄 테크 가부시키가이샤 | Flake-like silver powder, conductive paste, and method for producing flake-like silver powder |
| US11569538B2 (en) | 2014-06-16 | 2023-01-31 | The Regents Of The University Of California | Hybrid electrochemical cell |
| US10847852B2 (en) | 2014-06-16 | 2020-11-24 | The Regents Of The University Of California | Hybrid electrochemical cell |
| JP2018501644A (en) * | 2014-11-18 | 2018-01-18 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | Porous interconnected corrugated carbon based network (ICCN) composites |
| US11810716B2 (en) | 2014-11-18 | 2023-11-07 | The Regents Of The University Of California | Porous interconnected corrugated carbon-based network (ICCN) composite |
| JP2021108376A (en) * | 2014-11-18 | 2021-07-29 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニアThe Regents Of The University Of California | Porous interconnected wavy carbon-based network (ICCN) composite material |
| US10734167B2 (en) | 2014-11-18 | 2020-08-04 | The Regents Of The University Of California | Porous interconnected corrugated carbon-based network (ICCN) composite |
| JP2017110298A (en) * | 2015-12-14 | 2017-06-22 | ローム アンド ハース エレクトロニック マテリアルズ エルエルシーRohm and Haas Electronic Materials LLC | Environmentally friendly stable catalyst for electroless metalization of printed circuit board and through-hole |
| JP2017110297A (en) * | 2015-12-14 | 2017-06-22 | ローム アンド ハース エレクトロニック マテリアルズ エルエルシーRohm and Haas Electronic Materials LLC | Environmentally friendly stable catalyst for electroless metalization of printed circuit board and through-hole |
| JP2017110296A (en) * | 2015-12-14 | 2017-06-22 | ローム アンド ハース エレクトロニック マテリアルズ エルエルシーRohm and Haas Electronic Materials LLC | Environmentally friendly stable catalyst for electroless metalization of printed circuit board and through-hole |
| US11891539B2 (en) | 2015-12-22 | 2024-02-06 | The Regents Of The University Of California | Cellular graphene films |
| US10655020B2 (en) | 2015-12-22 | 2020-05-19 | The Regents Of The University Of California | Cellular graphene films |
| US11118073B2 (en) | 2015-12-22 | 2021-09-14 | The Regents Of The University Of California | Cellular graphene films |
| US10892109B2 (en) | 2016-01-22 | 2021-01-12 | The Regents Of The University Of California | High-voltage devices |
| US11842850B2 (en) | 2016-01-22 | 2023-12-12 | The Regents Of The University Of California | High-voltage devices |
| US11062855B2 (en) | 2016-03-23 | 2021-07-13 | The Regents Of The University Of California | Devices and methods for high voltage and solar applications |
| US11961667B2 (en) | 2016-03-23 | 2024-04-16 | The Regents Of The University Of California | Devices and methods for high voltage and solar applications |
| US11097951B2 (en) | 2016-06-24 | 2021-08-24 | The Regents Of The University Of California | Production of carbon-based oxide and reduced carbon-based oxide on a large scale |
| US11791453B2 (en) | 2016-08-31 | 2023-10-17 | The Regents Of The University Of California | Devices comprising carbon-based material and fabrication thereof |
| US10938021B2 (en) | 2016-08-31 | 2021-03-02 | The Regents Of The University Of California | Devices comprising carbon-based material and fabrication thereof |
| US11133134B2 (en) | 2017-07-14 | 2021-09-28 | The Regents Of The University Of California | Simple route to highly conductive porous graphene from carbon nanodots for supercapacitor applications |
| US10938032B1 (en) | 2019-09-27 | 2021-03-02 | The Regents Of The University Of California | Composite graphene energy storage methods, devices, and systems |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2005199267A (en) | Metal carrier and method for manufacturing the same | |
| Wu et al. | NiFe2O4 nanoparticles/NiFe layered double-hydroxide nanosheet heterostructure array for efficient overall water splitting at large current densities | |
| Chandra et al. | Controlled synthesis of CuS/TiO2 heterostructured nanocomposites for enhanced photocatalytic hydrogen generation through water splitting | |
| Wang et al. | Surface roughening of nickel cobalt phosphide nanowire arrays/Ni foam for enhanced hydrogen evolution activity | |
| Wang et al. | Quantum yield of formaldehyde formation in the presence of colloidal TiO2-based photocatalysts: effect of intermittent illumination, platinization, and deoxygenation | |
| Wang et al. | Surface engineered CuO nanowires with ZnO islands for CO2 photoreduction | |
| JP4564263B2 (en) | Ultrafine metal particle-containing photocatalyst and method for producing the same | |
| Ghoreishian et al. | γ-Radiolysis as a highly efficient green approach to the synthesis of metal nanoclusters: a review of mechanisms and applications | |
| JP2007098563A (en) | Nanostructure and method for producing nanostructure | |
| Lim et al. | Silver/titania nanocomposite-modified photoelectrodes for photoelectrocatalytic methanol oxidation | |
| Zimbone et al. | Sb-doped titanium oxide: A rationale for its photocatalytic activity for environmental remediation | |
| Solmaz | Gold‐supported activated NiZn coatings: hydrogen evolution and corrosion studies | |
| WO2005056222A1 (en) | Metal nanocolloidal liquid, method for producing metal support and metal support | |
| JP6767202B2 (en) | Metal-containing nanoparticles-supported electrode and carbon dioxide reduction device | |
| JP2015188808A (en) | Oxidation-reduction catalyst, electrode material, electrode, solar battery, membrane battery assembly for fuel battery, and fuel battery | |
| JP5292764B2 (en) | Cathode catalyst for fuel cell, production method and immobilization method thereof, and fuel cell | |
| JP2004332028A (en) | Ternary metallic colloid having three layer core/shell structure and method for producing ternary metallic colloid | |
| Xu et al. | Electrocatalytic dechlorination of florfenicol using a Pd-loaded on blue TiO2 nanotube arrays cathode | |
| KR101237449B1 (en) | A method for preparing Pt thin film using electrospray deposition and Pt thin film formed by the method | |
| CN110330078B (en) | High-efficiency long-life three-dimensional structure antimony-doped tin oxide electrode | |
| JP5001010B2 (en) | Method for producing noble metal particulate carrier | |
| JP5674553B2 (en) | Ozone decomposition removal catalyst, method for producing the same, and ozone decomposition removal method using the same | |
| KR101000475B1 (en) | Preparation method of transition metal/carbon nanotubes nanocomposite by electroless deposition | |
| JP2005169333A (en) | Metal nano-colloid liquid, carrier material, manufacturing method for metal carrier and metal carrier | |
| JP4647906B2 (en) | Method for producing metal carrier and metal carrier. |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20071001 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20091027 |
|
| A761 | Written withdrawal of application |
Free format text: JAPANESE INTERMEDIATE CODE: A761 Effective date: 20100312 |